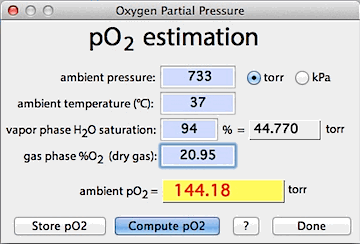
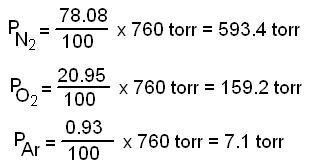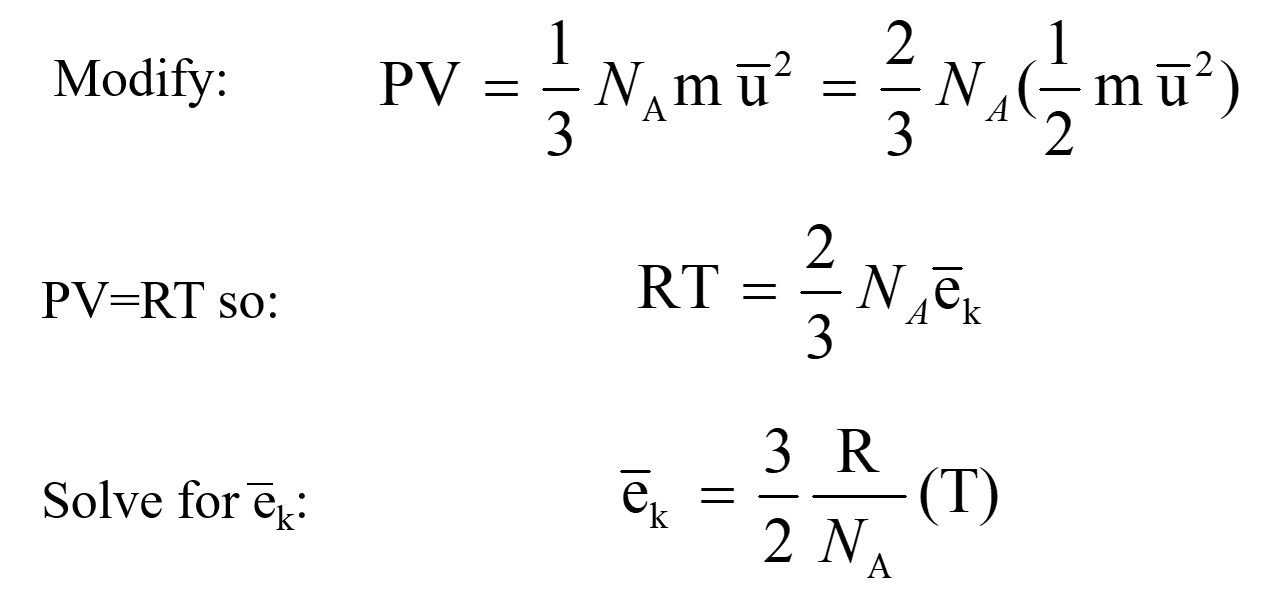![AP chemistry: partial pressures]How do I calculate the partial pressure and the volume of the jar? : r/HomeworkHelp AP chemistry: partial pressures]How do I calculate the partial pressure and the volume of the jar? : r/HomeworkHelp](https://preview.redd.it/4xvi44skj3y51.jpg?auto=webp&s=6c00aa66deba192a88060dcfb5679f4718180123)
AP chemistry: partial pressures]How do I calculate the partial pressure and the volume of the jar? : r/HomeworkHelp
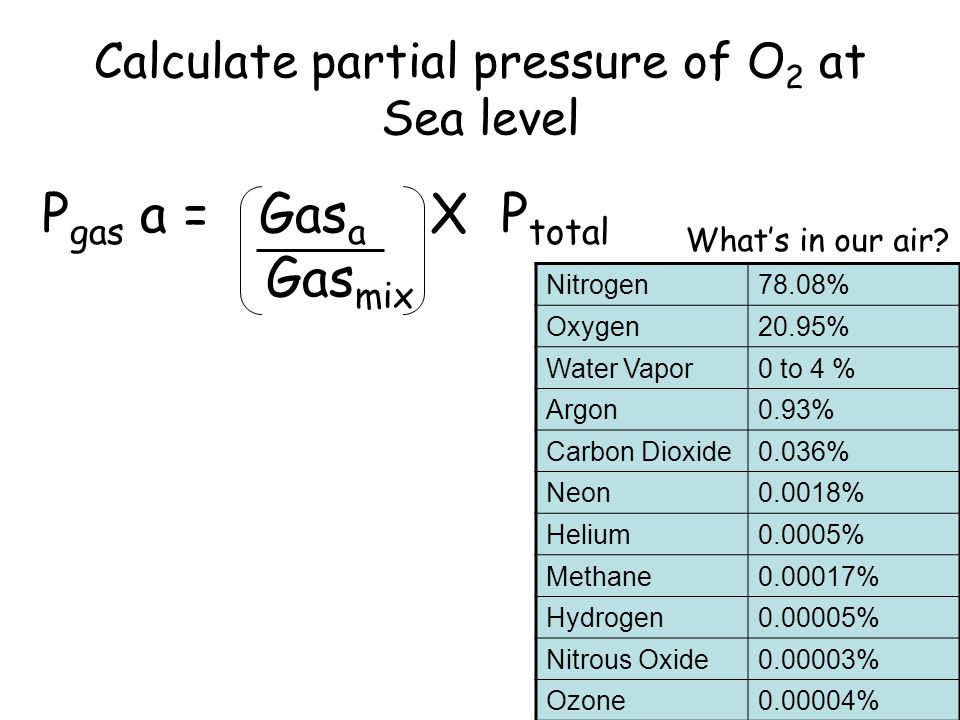
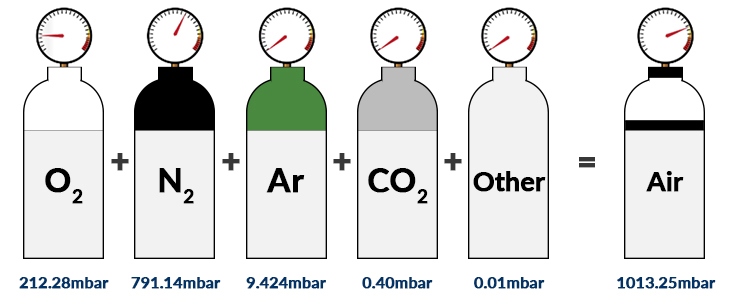
A mixture of 40 g of oxygen and 40 g of helium has a total pressure of 0.9 atm. The partial pressure of oxygen is (1) 0.5 atm (2) 0.1 atm (3) 09 atm (4) 0.2 atm